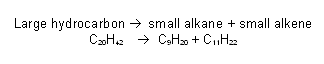Octane numbers
- The octane number of a fuel is based on how fuels auto ignite, under compression.
- It is based on the scale where isooctane is 100 (minimal ignition under pressure) and heptane is 0 (easily ignites under pressure).
- For example, a petrol with an octane number of 92 has the same knock as a mixture of 92% isooctane and 8% heptane.
- Octane rating decreases with an increase in the carbon chain length.
- Octane ratings increase with carbon chain branching.
- Octane ratings increase in aromatics with same number of carbons.
- It is important to know octane numbers for petrol because the auto ignition of fuels causes a ōknockingö effect in petrol engines. This is where it ignites twice; once due to the high pressure and again when the spark ignites the petrol. This causes the car engine to be less efficient and it can also damage the engine.
- However, diesel engines rely on this knocking effect, as they have no spark plugs and rely on the effect of compression to make their fuel/air mixtures ignite.
How octane rating of a fuel can be increased
- There are two ways to increase the octane number of a fuel. One is to put special additives into the fuel which discourage auto ignition, and the other is to blend high-octane fuels in with the ordinary petrol.
Anti-knocking additives
- Anti-knocking additives are substances which reduce the tendency of a fuel to auto-ignite, and so increase the octane number.
- Since the 1920Æs, small amounts of lead compounds have been used as economical and effective anti-knock additives.
- Concern over the environmental effects of leaded petrol has lead to a gradual phasing out of leaded petrol.
- Petrol companies have focused their attention on refining and blending to get high octane ratings.
The right kind of alkane
- The best kind of alkane for fuels is not the most plentiful in crude oil.
- The refinery ōdoctorsö the fuel for our needs.
- The shorter the alkane chain, the higher the octane number. However, the volatility of the hydrocarbon increases with a decrease in the chain length.
- The short alkane chains have to be mixed in petrol to lower their volatility and higher the octane number of the petrol.
- The other factor that affects the octane number is the degree of branching in the alkane chain. The more branched the alkane is, the higher the octane number.
- Crude oil contains both straight-chain and branched alkanes, but it does not contain enough branched alkanes to give it a high octane number.
- Chemists have solved this problem; increasing the octane numbers by isomerism, reforming and cracking.
Isomerism
- Isomerization involves heating up straight chained alkanes in the presence of suitable catalysts, so that the chains break.
- When the chains join together again, they are more likely to be branched.
- Oil refineries do this on a large scale with C6H14 and C5H12.

- The isomerization reaction is reversible and higher temperatures favour the production of straight chained isomers. However the temperature cannot be too low or the reaction would take too long, so a compromise is made between yield and rate of reaction. The temperature used is 250oC.
- The catalyst involved in the isomerization is platinum; the products are then passed over a form of zeolite, which acts as a molecular sieve, separating the straight chains from the branched chains.
Reforming
- Reforming is where alkanes are converted to cyclo-alkanes and cyclo alkanes to aromatic hydrocarbons.
- Substances such as hexane (which are common in crude oil) are converted to cyclo-hexane. Hydrogen is also produced, which is valuable.
Cracking
- Cracking is one of the most important reactions in the petroleum industry.
- Cracking involves breaking up large alkanes which have too many carbons for petrol into shorter chains that can be used in petrol.
- These shorter chains are often branched, which gives them a higher octane number.
- Cracking also helps to solve the supply and demand problem. Some fractions are abundant in the crude oil, but are not needed. However, other fuels are in great demand but are not common in the crude oil.
- Most of the cracking is done by heating up the fuel in the presence of a catalyst. This process is referred to as cat cracking. The fuel that is heated can be kerosene, diesel oil, or any other heavier fuels from the residue.
- Zeolites are once again used in this process, as they act as excellent catalysts. Zeolite Y is used to crack the fuel, as it particularly effective at producing fuels with a high octane rating.
- As well as the shorter chained alkanes, alkenes are also formed. These are more often branched and are important in the petrochemicals industry.
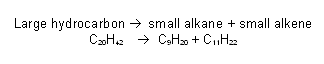
- One problem with cracking is that coke (carbon) tends to form on the catalyst surface, and so the catalyst eventually becomes inactive. To overcome this, the catalyst is circulated with the products and regenerated after each cycle.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home