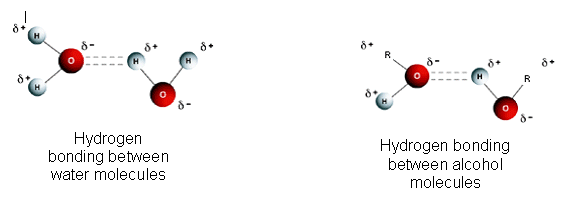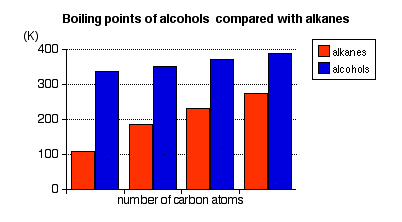Alcohols and Ethers
- All alcohols contain an ľOH group, for example ethanol. Alcohols have similar chemical properties.
- All ethers contain an ľOľ group, for example methoxymethane. Ethers have similar chemical properties.
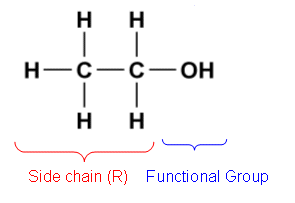
- Groups such as the ľOH and ľOľ which are substituted in a hydrocarbon chain are called functional groups.
- They dominate the chemistry of a molecule and give it characteristic properties. The hydrocarbon chain is the unreactive part of the molecule and is called the side chain and given the symbol R.
Alcohols
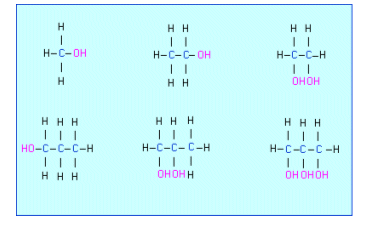
- Alcohols are hydrocarbon chains with an ľOH group replacing an ľH atom.
- They are named from the parent alkane by dropping the ô-eö from the end of the alkane and replacing it with an ô-olö.
- For alcohols containing more than two carbon atoms, isomeric compounds are possible. To distinguish between them it is necessary to label the position of the OH group.
- As with alkanes the number is the shortest that it can be, so instead of butan-4-ol, it would be butan-1-ol.
- Polyhydric alcohols have more than one ľOH group and are named diol for 2 and triol for 3.
- Compounds with an alcohol attached directly to a benzene ring are called phenols.
Physical properties of alcohols
- Alcohols are like water, apart from one of the hydrogen atoms has been replaced by an alkyl group.

- Water molecules and alcohol molecules are polar (has a negative and positive end) because of their O-H bond. The strong attractive forces between the molecules are known as hydrogen bonds.
- Hydrogen bonds are weaker than covalent bonds, but stronger than other attractive forces between covalent molecules (for example, van der waalĺs forces).
- The hydrogen atoms within the O-H group are slightly positive, because the electrons are pulled away from them by the highly electronegative oxygen atoms.
- The oxygen atoms are slightly negative, because the electrons are closer to them.
- The positive hydrogen atoms and negative oxygen atoms in neighbouring molecules are attracted to each other forming a hydrogen bond approximately 5% as strong as a covalent bond.
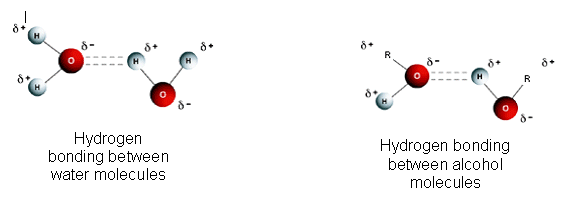
- It is because of the strong hydrogen bond in alcohols that makes them have a higher boiling point than their corresponding alkanes which have a similar Mr.
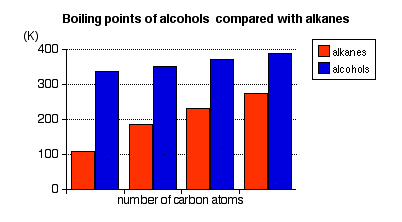
- The solubility of alcohols decreases as you increase the size of the molecule.
- When an alcohol is added to water, the hydrogen bonds in the water and alcohol have to be broken in order for them to mix.
- Breaking bonds requires energy; however (in smaller molecules) when the hydrogen bonds are reformed between the alcohol and water molecules energy is released, and this compensates for the initial energy needed to break the bonds.
- In larger molecules, the hydrocarbon tail of the molecule needs to fit in between the water molecules, and so it breaks more hydrogen bonds between the molecules.
- The tail cannot form hydrogen bonds, so the hydrogen bonds are replaced by van der waalĺs forces. These forces are much weaker, and so not as much energy is released when they form; not all of the energy needed to break the hydrogen bonds has been compensated for. So larger molecules do not dissolve as easily in water in comparison to smaller molecules.
Ethers
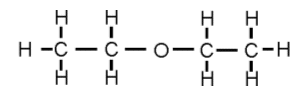
- Ethers are derived from alkanes by replacing a H atom for a alkoxyl group (-OR). For example:
- Ethoxyethane is formed from butane, but two of the hydrogen atoms have been replaced by an oxyen atom.
To name the ether the longest hydrocarbon chain is placed at the end and an oxy is placed before it (oxyalkane).
- Ethers are derived from water, in that they have the central oxygen atom, with chains of hydrocarbons coming from each side.
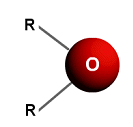
- There are no Hydrogen bonds holding together the structure of ethers; only relatively weak forces hold the slightly polar molecules together.
- The boiling points of ethers are much the same as their counterpart alkanes.
- Lower ethers are extremely volatile and dangerously flammable.
- Ethers are only slightly soluble in water, but mix well with other non-polar molecules such as alkanes.
Useful books for revision:
Revise AS Chemistry for Salters (Written by experienced examiners and teachers of Salter's chemistry)
Revise AS Chemistry for Salters (OCR) (Salters Advanced Chemistry)
Home